Health Canada announces decision to approve submission from global pharmaceutical company Dr. Reddy’s Laboratories
Published Apr 28, 2026 • Last updated 20 minutes ago • 2 minute read

Canada is the first G7 nation to allow a generic version of a semaglutide injection after a pharmaceutical equivalent of the popular Ozempic drug received regulatory approval.
Advertisement 2
THIS CONTENT IS RESERVED FOR SUBSCRIBERS ONLY
Subscribe now to read the latest news in your city and across Canada.
- Unlimited online access to articles from across Canada with one account.
- Get exclusive access to the Toronto Sun ePaper, an electronic replica of the print edition that you can share, download and comment on.
- Enjoy insights and behind-the-scenes analysis from our award-winning journalists.
- Support local journalists and the next generation of journalists.
- Daily puzzles including the New York Times Crossword.
SUBSCRIBE TO UNLOCK MORE ARTICLES
Subscribe now to read the latest news in your city and across Canada.
- Unlimited online access to articles from across Canada with one account.
- Get exclusive access to the Toronto Sun ePaper, an electronic replica of the print edition that you can share, download and comment on.
- Enjoy insights and behind-the-scenes analysis from our award-winning journalists.
- Support local journalists and the next generation of journalists.
- Daily puzzles including the New York Times Crossword.
REGISTER / SIGN IN TO UNLOCK MORE ARTICLES
Create an account or sign in to continue with your reading experience.
- Access articles from across Canada with one account.
- Share your thoughts and join the conversation in the comments.
- Enjoy additional articles per month.
- Get email updates from your favourite authors.
THIS ARTICLE IS FREE TO READ REGISTER TO UNLOCK.
Create an account or sign in to continue with your reading experience.
- Access articles from across Canada with one account
- Share your thoughts and join the conversation in the comments
- Enjoy additional articles per month
- Get email updates from your favourite authors
Article content
Health Canada announced its decision on Tuesday to approve the submission filed by the global pharmaceutical company Dr. Reddy’s Laboratories, which is based out of Hyderabad, India, and operates a U.S. headquarters in New Jersey.
Article content
Article content
They said eight other generic versions of the drug are in the queue to be approved by regulators and that the “complex” synthetic products are “pharmaceutically equivalent to the brand-name biologic drug.
“Health Canada’s review ensures that differences between these products do not affect the safety, efficacy or quality of the drug,” the department’s news bulletin said. “The availability of generic drugs is expected to have a positive impact in Canada, including potential cost savings for patients and the health-care system.
“As it does with all drugs authorized in Canada, Health Canada will continue to monitor the safety and effectiveness of all generic semaglutide products.”
RECOMMENDED VIDEO
We apologize, but this video has failed to load.
Generic drug costs drop with more competition
The drug is used for the weekly treatment of Type 2 diabetes in adult patients who need to manage their blood-sugar levels, according to Health Canada, which added that “many” generic drugs are about 45% to 90% cheaper than brand-name versions.
Advertisement 3
Article content
By comparison, a prescription for Novo Nordisk’s Ozempic brand – which has gained attention for being prescribed as an off-label method to help with weight loss – can cost Canadians hundreds of dollars a month, according to The Canadian Press.
The cost of generic versions will depend on how many are approved and end up on the market with the pan-Canadian Pharmaceutical Alliance pricing structure listing a single-source product potentially costing about 75% to 85% of the price of the brand-name product.
Dual-source products can be priced at 50% of the cost of a brand-name medication, while multi-source products drop to 25% to 35% of the brand-name cost.
India gave the OK to two generic semaglutide drugs made by Dr. Reddy’s Laboratories in March after Ozempic’s patent expired there, according to The Canadian Press. That was after they submitted their product for review in Canada in 2024.
Read More
-

What is ‘Ozempic personality,’ and why does it make life feel ‘meh’?
-

If you aren’t losing weight with GLP-1 drugs, this may be one reason why
Article content
.png)
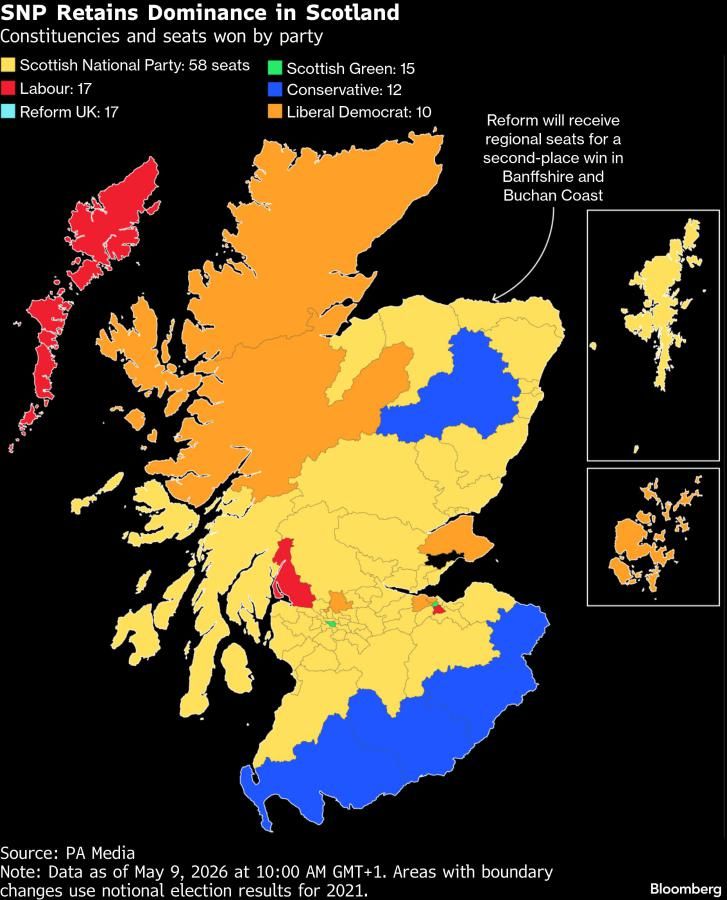
 1 week ago
17
1 week ago
17






























 Bengali (BD) ·
Bengali (BD) ·  English (US) ·
English (US) ·